UCSF Musculoskeletal Research Consortium (METRiCS)
Our work
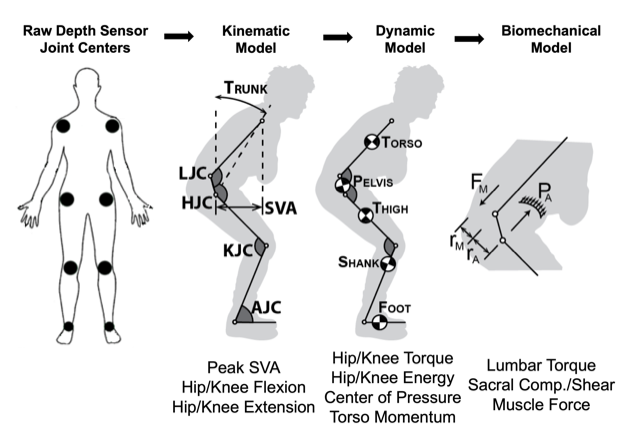
The UCSF Musculoskeletal Research Consortium, or METRiCS, seeks to foster basic, clinical, and translational research involving the neuromusculoskeletal system. METRiCS aspires to be a “research commons” where like-minded scientists can work across disciplines, campuses, Departments, and Schools to achieve innovative, collaborative solutions to vexing musculoskeletal questions. As an example, the UCSF international Center for Osseointegration Research, Education, and Surgery (iCORES) program leverages expertise in percutaneous osseointegration technology to better rehabilitate amputees, while developing implanted brain-computer interface (BCI) devices for patients with paralysis or amputation. Directed by Richard J. O’Donnell, M.D., METRiCS has enabled Jeannie F. Bailey, Ph.D. and Robert P. Matthew, Ph.D. the launch of the Amputee Safe Smart Implant Sensor Technology (ASSIST) program to track biomechanical function, maximize biomechanical ability, and improve the safety and predictability of return to maximal participation in civilian and military activities. Emerging METRiCS projects include colleagues in the Departments of Orthopaedic Surgery, Physical Therapy, Radiology, Surgery, Medicine, and Otolarynology/Head and Neck Surgery; in the Schools of Medicine and Dentistry; and at UCSF and UC Berkeley.
Current projects
METRiCS currently has a total of 19 research protocols approved by the UCSF Institutional Review Board (IRB), including 5 clinical trials for a variety of patient populations. Interested individuals can contact our program at METRiCSinfo@ucsf.edu. Our research is funded in part through four federal contracts and grants. We are thankful for the support of colleagues and funders at the Department of Defense Osseointegration Program; the Walter Reed National Military Medical Center; the Uniformed Services University; the Defense Advanced Research Projects Agency (DARPA); the Space and Naval Warfare Systems Command (SPAWAR); the U.S. Army Medical Research and Development Command (USAMRDC); the Congressionally Directed Medical Research Program (CDMRP); the Henry M. Jackson Foundation for the Advancement of Military Medicine (HJF); the Massachusetts Institute of Technology; Harvard Medical School; the Brigham and Women’s Hospital; the Johns Hopkins University; and the University of California, Berkeley.
IRB approved research protocols
- 18-25115 Neuroimaging-Based Objective Diagnostic Tool to Detect Subjective Tinnitus
- 21-33990 OPRA Implant System Premarket Approval and Related Studies
- 20-32576 A 2-year follow-up, multidisciplinary study focused on outcomes and experience with the OPRA Axor II coupling unit
- 20-31453 Phantom Limb Pain Multimodal Neuroimaging
- 20-30167 Osseointegration Quality Registry
- 19-28003 ASSIST: Amputee Safe Smart Implant Sensor Technology
- 18-25641 Transfemoral Amputation Osseointegration Study, TFAOS, Non-Research, HUD Only
- 18-25404 Transfemoral Amputation Osseointegration Study, TFAOS, Research
- 18-24966 Transhumeral e-OPRA, An osseoanchored percutaneous prosthesis study evaluating stable neural signal transmission in subjects with transhumeral amputations
- 18-24855 Incidence of amputees who may be candidates for an osseointegrated percutaneous prosthetic system
- 17-24173 Monitoring post-surgical postural kinematics in adult spinal deformity patients using inertial monitoring units (IMUs)
- 17-22291 Clinical assessment of spinal stability using motion capture
- 17-22205 Patient Preferences for Attributes of Risk and Benefit of Prosthetic Devices for Upper Limb Loss
- 17-21928 An osseointegrated transfemoral prosthesis offering long-term bidirectional efferent-afferent neural transmission (IDE)
- 16-19786 Transhumeral Amputation Osseointegration Study, TAOS
- 15-18025 Compressive osseointegration for distal femur reconstruction in giant cell tumor
- 15-16764 Osseointegrated percutaneous prosthetic system for transfemoral amputees; A prospective cohort study
- 12-4872 Assistive exoskeleton for activities of daily living
- 11-10515 Affordable devices to assess, assist, and resist upper limb motions
- 10-02939 Reconstruction of massive defects using the ComPreSs device for endoprosthetic fixation in the proximal femur, distal femur, proximal tibia and humerus
- 07-6506 Human-centered modelling and control of cooperative manipulation
- 04-10940 Analysis of human motion with an assistive knee orthotic
- 01-8261 Individualized models and assistive devices for the upper limbs
Clinical trials
- DARPA-W911NF-17-2-0043. An osseointegrated transfemoral prosthesis offering long-term bi-directional efferent-afferent neural transmission.
- W81XWH-17-2-0060, USAMRMC-BA160465. Transfemoral Amputee Osseointegration Study (TFAOS).
- DARPA-15-05-OpenBAA-FP-014. An osseo-neural transtibial prosthesis with efferent-afferent control.
- 1U19AR076737-01. UCSF Core Center for Patient-Centric Phenotyping in Chronic Low Back Pain.
- W81XWH-19-OPORP-CRA. Assessing biomechanical function and hip stabilizing muscle quality associated with transfemoral osseointegration.
Publications
- Zaid, M.B.; O'Donnell, R.J.; Potter, B.K.; and Forsberg, J.A.: Orthopaedic osseointegration: State of the art. J.Amer. Acad. Orthop. Surg., 27(22):e977-e985, 2019. doi: 10.5435/JAAOS-D-19-00016. PubMed PMID: 31181031.
- Zaid, M.B.; Wustrack, R.L.; Garibaldi, M.; Geiger, E.; Andaya, V.; and O’Donnell, R.J. Prospective study of percutaneous bone-anchored implants in transfemoral amputees: Brain-machine platform technology for external prosthetic control and feedback. Conf. Proc. IEEE Eng. Med. Biol. Soc., 2019 Mar;2019:13-16.
- Strony, J.; Brown, S.; Choong, P.; Ghert, M.; Jeys, L.; and O’Donnell, R.J.: Musculoskeletal infection in orthopaedic oncology: Assessment of the 2018 International Consensus Meeting on Musculoskeletal Infection. J. Bone Joint Surg., 101-A(20):e107, 2019. doi: 10.2106/JBJS.19.00182. PubMed PMID: 31626015.
- Bailey, J.F.; Matthew, R.P.; Seko, S.; Curran, P.; Chu, L.; Deviren, V.; Berven, S.H.; Burch, S.; and Lotz, J.C. Biomechanical changes in dynamic sagittal balance and lower limb compensatory strategies following realignment surgery in adult spinal deformity patients. Euro. Spine J., 28(5):905-913, 2019. doi: 10.1007/s00586-019-05925-2. Epub 2019 Mar 2. PubMed PMID: 30826876 (2019 ISSLS Prize).
- Bailey, J.F.; Matthew, R.P.; Seko, S.; Curran, P.; Chu, L.; Deviren, V.; Berven, S.H.; Burch, S.; and Lotz, J.C. Biomechanical changes in dynamic sagittal balance and lower limb compensatory strategies following realignment surgery in adult spinal deformity patients. Euro. Spine J., 2019. In press. (2019 ISSLS Prize)
- Matthew, R.P.; Seko, S.; Bailey, J.; Bajcsy, R.; and Lotz, J. Estimating sit-to-stand dynamcis using a single depth camera. IEEE J. Biomed. Health Inform., 2019. doi: 10.1109/JBHI.2019.2897245. Epub 2019 Feb 4. PubMed PMID: 30716057.
- Matthew, R.P.; Seko, S.; Bajcsy, R.; and Lotz, J. Kinematic and kinetic validation of an improved depth camera motion assessment system using rigid bodies. IEEE J. Biomed. Health Inform., 2018. doi: 10.1109/JBHI.2018.2872834. Epub 2018 Oct 1. PubMed PMID: 30281504.
- Kanieshi, D.; Matthew, R.P.; and Tomizuka, M. A sEMG classification framework with less training data. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2018(Jul):1680-1684, 2018. doi: 10.1109/EMBC.2018.8512623. PubMed PMID: 30440718.
- Seko, S.; Matthew, R.P.; Bajcsy, R.; and Lotz, J.C. A functional method for generating individualized spine models from motion-capture data. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2018(Jul):1485-1489, 2018. doi: 10.1109/EMBC.2018.8512580. PubMed PMID: 30440263.
- Matthew, R.P.; Seko, S.; Bailey, J.; Bajcsy, R.; and Lotz, J. Tracking kinematic and kinetic measures of sit to stand using an instrumented spine orthosis. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2018(Jul):1197-1201, 2018. doi: 10.1109/EMBC.2018.8512526. PubMed PMID: 30440257.
- Bailey, J.F.; Miller, S.L.; Khieu, K.; O’Neill, C.W.; Healey, R.M.; Coughlin, D.G.; Sayson, J.V.; Chang, D.G.; Hargens, A.R.; and Lotz, J.C. From the international space station to the clinic: How prolonged unloading may disrupt lumbar spine stability. Spine J., 18(1):7-14, 2018. doi: 10.1016/j.spinee.2017.08.261. Epub 2017 Sep 28. PubMed PMID: 28962911.
- Matthew, R.P.; Seko, S.; and Bajcsy, R. Fusing motion-capture and inertial measurements for improved joint state recovery: An application for sit-to-stand actions. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2017(Jul):1893-1896, 2017. doi: 10.1109/EMBC.2017.8037217. PubMed PMID: 29060261.
- Chang, D.G.; Healey, R.M.; Snyder, A.J.; Sayson, J.V.; Macias, B.R.; Coughlin D.G.; Bailey, J.F.; Prazynski, S.E.; Lotz, J.C.; and Hargens, A.R. Lumbar spine paraspinal muscle and intervertebral disc height changes in astronauts after long-duration spaceflight on the International Space Station. Spine, 41(24):1917-1924, 2016. doi: 10.1097/BRS.0000000000001873. PubMED PMID: 27779600.
- Goldman, L.H.; Morse, L.J.; O’Donnell, R.J.; and Wustrack, R.L. How often does spindle failure occur in compressive osseointegration endoprostheses for oncologic reconstruction? Clin. Orthop. Rel. Res., 474:1714-1723, 2016. doi: 10.1007/s11999-016-4839-7. Epub 2016 Apr 22. PubMed PMID: 27106130.
- Matthew, R.P.; Shia, V.; Venture, G.; and Bajcsy, R. Generating physically realistic kinematic and dynamic models from small data sets: An application for sit-to-stand actions. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2016(Aug):2173-2178, 2016. doi: 10.1109/EMBC.2016.7591160. PubMed PMID: 28268763.
- Goldman, L.H.; Morse L.J.; O’Donnell, R.J.; and Wustrack, R.L. How often does spindle failure occur in compressive osseointegration endoprostheses for oncologic reconstruction? Clin. Orthop. Rel. Res., 474:1714-1723, 2016. doi: 10.1007/s11999-016-4839-7. Epub 2016 Apr 22. PubMed PMID: 27106130.
- Matthew, R.P.; Mica, E.J.; Meinhold, W.; Loeza, J.A.; Tomizuka, M.; and Bajcsy, R. Initial investigation into the effect of an active/passive exoskeleton in hammer curl performance in healthy subjects. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2015(Aug):3607-3610, 2015. doi: 10.1109/EMBC.2015.7319173. Pub Med PMID: 26737073.
- Pedtke, A.C.; Wustrack, R.L.; Fang, A.S.; Grimer, R.J.; and O’Donnell, R.J.: Aseptic failure: How does the Compress® implant compare to cemented stems? Clin. Orthop. Rel. Res., 470:735-742, 2012. doi: 10.1007/s11999-011-2159-5. Epub 2011 Nov 2. PubMed PMID: 22045069.
- O’Donnell, R.J.: Compressive osseointegration of tibial implants in primary cancer reconstruction. Clin. Orthop. Rel. Res., 467:2807-2812, 2009. doi: 10.1007/s11999-009-0986-4. Epub 2009 Aug 4. PubMed PMID: 19653050.
- O’Donnell, R.J.: Compressive osseointegration of modular endoprostheses. Current Opinion Orthop., 18:590-603, 2007. doi: 10.1097/BCO.0b013e3282f0dafc.
- Avedian, R.S.; Goldsby, R.E.; Kramer, M.J.; and O’Donnell, R.J.: Effect of chemotherapy on initial compressive osseointegration of tumor endoprostheses. Clin. Orthop. Rel. Res., 459:48-53, 2007. doi: 10.1097/BLO.0b013e3180514c66. PubMed PMID: 17545758.